 The charge is not on any one atom but rather on the group of atoms as a whole. Using the chlorine family of polyatomic ions as a model predict the name of the Bro l- ion. Write out the name and formula of the ions including their charges. Genetic Mutations Pogil Answer Key Quizzma. Using the chlorine family of polyatomic ions as a model predict the name of the bro l ion. Polyatomic Ions Answer Key Pogil Polyatomic Ion Scientific Method Worksheet Worksheet Template. POGIL is an acronym for Process Oriented Guided Inquiry Learning. Polyatomic ions pogil activity answer key. Macromolecules Review Chart With Answers Macromolecules Biology Worksheet Photosynthesis Worksheet Find this Pin and more on af by Fanny sot. Bromine forms polyatomic ions with structures similar to those of chlorine. In other words, the ignoring was a perfectly valid thing to do.Polyatomic Ions Pogil Answer Key Model 2 Answers To Chemical Bonding Pogil. Substituting into a quadratic solver gives: If you decided that the additional amount of silver ion CANNOT be ignored, you would have this: To two significant digits, the bromide concentration is 1.1 x 10¯ 11 mol/L 
This value is small compared to 0.050 M, it can be ignored. The solution will also contain additional Ag +, due to the dissociation of the AgBr. The solution already contains 0.050 M Ag + from the dissociation of the AgNO 3. Calculate the molar solubility of AgBr in 0.050 M AgNO 3(aq) at 25 ☌. Problem #6: The K sp of AgBr is 5.4 x 10¯ 13 at 25 ☌. Problem #5 - Part 3: Calculate the molar solubility of Cr(OH) 3 in a solution buffered at pH = 11.00 Q is a 'reaction quotient.' You might have to look that up. Problem #5 - Part 2: 4.0 x 10 -4 g of NaOH is added. 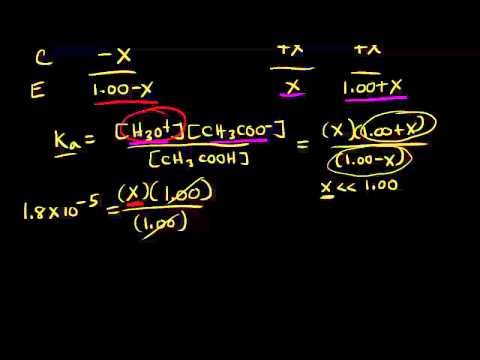
Problem #5: 1.1 x 10 -4 g of Cr(OH) 3 is added to 120. Problem #4: What is the maximum concentration of Mg 2+ ion that can remain dissolved in a solution that contains 0.7147 M NH 3 and 0.2073 M NH 4Cl? (K sp for Mg(OH) 2 is 1.2 x 10¯ 11 K b for NH 3 is 1.77 x 10¯ 5)ġ) Use the acid base data supplied to calculate :Ģ) Use the K sp expression to calculate the : Since we were asked for the moles of silver chromate that would disolve in 1.00 L, the final answer is: Since there is a 2:1 ratio between the moles of aqueous silver ion and the moles of silver chromate that dissolved, 1.5 x 10 -5 M is the molar solubility of Ag 2CrO 4 in 0.010 M K 2CrO 4 solution. K sp for Ag 2CrO 4 = 9.0 x 10 -12.ġ) Concentration of dichromate ion from potassium chromate: 
Problem #3: Calculate the number of moles of Ag 2CrO 4 that will dissolve in 1.00 L of 0.010 M K 2CrO 4 solution. Note how zinc hydroxide would precipitate even when the solution is slightly acidic. X = 5.91657 x 10 -8 M (I kept a few guard digits.) Problem #2: Calculate the pH at which zinc hydroxide just starts to precipitate from a 0.00857 M solution of zinc nitrate. 
Substituting into the K sp expression:Īny sodium hydroxide solution greater than 0.33 M will reduce the to less than 1.1 x 10¯ 10 M. What minimum OH¯ concentration must be attained (for example, by adding NaOH) to decrease the Mg 2+ concentration in a solution of Mg(NO 3) 2 to less than 1.1 x 10¯ 10 M? Problem #1: The solubility product of Mg(OH) 2 is 1.2 x 10¯ 11. The Common Ion Effect - Problems 1 - 10 The Common Ion Effect 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
